Determining Electron-Group & Molecular Geometry Chart
ADVERTISEMENT
One Page Lesson: Determining Electron-Group & Molecular Geometry
The repulsive forces between bonding and non-bonding electrons determine the three-dimensional geometry of the
“groups” of electrons around a central atom. Because the negative charges repel one another, the electron groups arrange
themselves so they are as far apart as possible. This is the “Valence Shell Electron Pair Repulsion” (VSEPR) Theory.
Thus, the “Electron Group” geometry of each central atom in a structure can be determined by simply counting the
number of “groups” of electrons around the atom, then considering how those groups would arrange themselves to be as
far apart as possible. A “group” of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons.
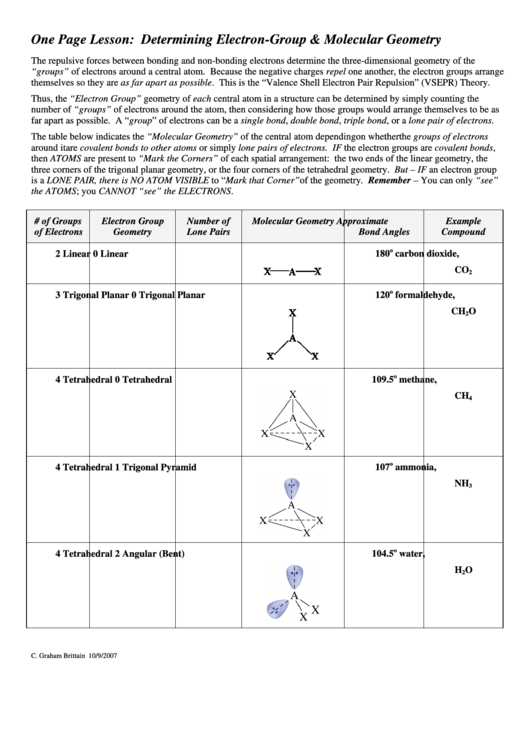
The table below indicates the “Molecular Geometry” of the central atom depending on whether the groups of electrons
around it are covalent bonds to other atoms or simply lone pairs of electrons. IF the electron groups are covalent bonds,
then ATOMS are present to “Mark the Corners” of each spatial arrangement: the two ends of the linear geometry, the
three corners of the trigonal planar geometry, or the four corners of the tetrahedral geometry. But – IF an electron group
is a LONE PAIR, there is NO ATOM VISIBLE to “Mark that Corner”of the geometry. Remember – You can only “see”
the ATOMS; you CANNOT “see” the ELECTRONS.
# of Groups
Electron Group
Number of
Molecular Geometry
Approximate
Example
of Electrons
Geometry
Lone Pairs
Bond Angles
Compound
o
2
Linear
0
Linear
180
carbon dioxide,
CO
2
o
3
Trigonal Planar
0
Trigonal Planar
120
formaldehyde,
CH
O
2
o
4
Tetrahedral
0
Tetrahedral
109.5
methane,
CH
4
o
4
Tetrahedral
1
Trigonal Pyramid
107
ammonia,
NH
3
o
4
Tetrahedral
2
Angular (Bent)
104.5
water,
H
O
2
C. Graham Brittain
10/9/2007
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1