Eas 6560 Isotope Geochemistry Worksheet With Answers - 2015
ADVERTISEMENT
EAS 6560 Isotope Geochemistry
Problem Set 1 Solution
Due Feb. 3, 2015
1.
a.
How
many
moles
of
Nd
(AW
=
144.24u;
u
is
unified
atomic
mass
units)
are
there
in
50
g
of
Nd
O
(AW
of
2
3
oxygen
i s
1 5.999u)?
336.47 is the molecular weight of Nd
O
, but one mole of Nd
O
contains two
2
3
2
3
moles of Nd. We have 50/336.47= 0.149 moles of Nd
O
and twice that, or
2
3
0.297 moles of Nd.
b .
H ow
m any
a toms
o f
N d
i n
t he
a bove?
23
23
0.297 × 6.022 × 10
= 1.799 x 10
atoms
2.
Using
the
equation
and
values
for
the
liquid
drop
model
given
in
the
notes,
predict
the
4
56
238
binding
e nergy
p er
n ucleon
f or
He,
Fe,
a nd
U
( ignore
e ven-‐odd
e ffects).
14A-‐13A^2/3-‐18.1I^2/4A-‐.58Z^2/A^0.33
A
Z
N
I
E
E
/nucleon
total
b
4
He
4
2
2
0
21.78054
5.44513
56
Fe
56
26
30
4
489.9410
8.748948
238
U
238
92
146
54
1985.1101
8.340799
3.
How
many
stable
nuclides
are
there
with
N
=
82?
List
them.
How
many
with
N
=
83?
List
them.
Why
the
difference?
144
142
141
140
139
138
136
N=82:
Sm,
Nd,
Pr,
Ce,
La,
Ba,
Xe
143
N-83:
Nd
82 is a magic number (also even).
4.
A
certain
radionuclide
emits
radiation
at
the
rate
of
15.0µW
at
one
instant
of
time
and
at
1.0
µW
(microwatts)
o ne
h our
l atter.
W hat
i s
i ts
h alf-‐life?
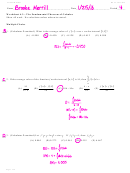
The activity, i.e., decays per unit time is -dN/dt. From the basic equation of
radioactive decay, -dN/dt = λN. Therefore:
λ N
dN / dt
N
=
=
0
0
0
λ N
dN / dt
N
1hour
1hour
1hour
⎛
⎞
N
Also
− λ t = ln
⎝ ⎜
⎠ ⎟
N
0
Substituting 1.0/15.0 for N/N
, 1 hour for t , we can solve for λ and convert that
0
-1
to half life, which works out to be: 0.256 hr (15.4 min) (λ = 2.708 hr
)
1
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3