Common Cations, Anions, Acids, Salts And Hydrate Nomenclature
ADVERTISEMENT
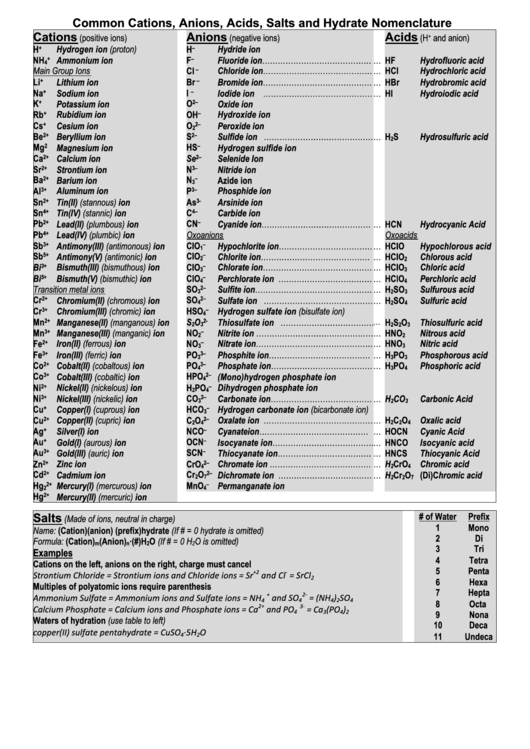
Common Cations, Anions, Acids, Salts and Hydrate Nomenclature
Cations
Anions
Acids
(positive ions)
(negative ions)
(H
and anion)
+
H
Hydrogen ion (proton)
H
Hydride ion
+
–
NH
Ammonium ion
F
Fluoride ion…………………………………… … HF
Hydrofluoric acid
+
–
4
Main Group Ions
Cl
Chloride ion…………………………………… … HCl
Hydrochloric acid
–
Li
Lithium ion
Br
Bromide ion…………………………………… … HBr
Hydrobromic acid
+
–
Na
Sodium ion
I
Iodide ion …………………………………… … HI
Hydroiodic acid
+
–
K
+
Potassium ion
O
2–
Oxide ion
Rb
+
Rubidium ion
OH
–
Hydroxide ion
Cs
Cesium ion
O
Peroxide ion
+
2–
2
Be
Beryllium ion
S
Sulfide ion …………………………………… … H
S
Hydrosulfuric acid
2+
2–
2
Mg
Magnesium ion
HS
Hydrogen sulfide ion
2
–
Ca
Calcium ion
Se
Selenide Ion
2+
2–
Sr
Strontium ion
N
Nitride ion
2+
3–
Ba
2+
Barium ion
N
–
Azide ion
3
AI
3+
Aluminum ion
P
3–
Phosphide ion
Sn
2+
Tin(II) (stannous) ion
As
3-
Arsinide ion
Sn
Tin(IV) (stannic) ion
C
Carbide ion
4+
4–
Pb
Lead(II) (plumbous) ion
CN
Cyanide ion…………………………………… … HCN
Hydrocyanic Acid
2+
–
Pb
Lead(IV) (plumbic) ion
Oxoanions
Oxoacids
4+
Sb
Antimony(III) (antimonous) ion
CIO
Hypochlorite ion……………………………… … HCIO
Hypochlorous acid
3+
–
1
Sb
Antimony(V) (antimonic) ion
CIO
Chlorite ion…………………………………… … HCIO
Chlorous acid
5+
–
2
2
Bi
Bismuth(III) (bismuthous) ion
CIO
Chlorate ion…………………………………… … HCIO
Chloric acid
3+
–
3
3
Bi
Bismuth(V) (bismuthic) ion
ClO
Perchlorate ion ……………………………… … HClO
Perchloric acid
5+
–
4
4
Transition metal ions
SO
Sulfite ion……………………………………… … H
SO
Sulfurous acid
2–
3
2
3
Cr
Chromium(II) (chromous) ion
SO
Sulfate ion …………………………………… … H
SO
Sulfuric acid
2+
2–
4
2
4
Cr
3+
Chromium(III) (chromic) ion
HSO
–
Hydrogen sulfate ion (bisulfate ion)
4
Mn
2+
Manganese(II) (manganous) ion
S
O
2-
Thiosulfate ion ………………………………
…
H
S
O
Thiosulfuric acid
2
3
2
2
3
Mn
3+
Manganese(III) (manganic) ion
NO
–
Nitrite ion ……………………………………… … HNO
Nitrous acid
2
2
Fe
Iron(II) (ferrous) ion
NO
Nitrate ion……………………………………… … HNO
Nitric acid
2+
–
3
3
Fe
Iron(III) (ferric) ion
PO
Phosphite ion………………………………… … H
PO
Phosphorous acid
3+
3–
3
3
3
Co
Cobalt(II) (cobaltous) ion
PO
Phosphate ion………………………………… … H
PO
Phosphoric acid
2+
3–
4
3
4
Co
Cobalt(III) (cobaltic) ion
HPO
(Mono)hydrogen phosphate ion
3+
2–
4
Ni
Nickel(II) (nickelous) ion
H
PO
Dihydrogen phosphate ion
2+
–
2
4
Ni
Nickel(III) (nickelic) ion
CO
Carbonate ion………………………………… … H
CO
Carbonic Acid
3+
2–
3
2
3
Cu
+
Copper(l) (cuprous) ion
HCO
–
Hydrogen carbonate ion (bicarbonate ion)
3
Cu
Copper(II) (cupric) ion
C
O
Oxalate ion …………………………………… … H
C
O
Oxalic acid
2+
2–
2
4
2
2
4
Ag
Silver(I) ion
NCO
Cyanate ion…………………………………… … HOCN
Cyanic Acid
+
–
Au
Gold(I) (aurous) ion
OCN
Isocyanate ion………………………………… … HNCO
Isocyanic acid
+
–
Au
Gold(III) (auric) ion
SCN
Thiocyanate ion……………………………… … HNCS
Thiocyanic Acid
3+
–
Zn
Zinc ion
CrO
Chromate ion ………………………………… … H
CrO
Chromic acid
2+
2–
4
2
4
Cd
Cadmium ion
Cr
O
Dichromate ion ……………………………… … H
Cr
O
(Di)Chromic acid
2+
2–
2
7
2
2
7
Hg
Mercury(I) (mercurous) ion
MnO
Permanganate ion
2+
–
2
4
Hg
Mercury(II) (mercuric) ion
2+
# of Water
Prefix
Salts
(Made of ions, neutral in charge)
1
Mono
Name: (Cation)(anion) (prefix)hydrate (If # = 0 hydrate is omitted)
2
Di
Formula: (Cation)
(Anion)
·(#)H
O (If # = 0 H
O is omitted)
m
n
2
2
3
Tri
Examples
4
Tetra
Cations on the left, anions on the right, charge must cancel
5
Penta
+2
‐
Strontium Chloride = Strontium ions and Chloride ions = Sr
and Cl
= SrCl
2
6
Hexa
Multiples of polyatomic ions require parenthesis
7
Hepta
+
2‐
Ammonium Sulfate = Ammonium ions and Sulfate ions = NH
and SO
= (NH
)
SO
4
4
4
2
4
8
Octa
2+
3‐
Calcium Phosphate = Calcium ions and Phosphate ions = Ca
and PO
= Ca
(PO
)
4
3
4
2
9
Nona
Waters of hydration (use table to left)
10
Deca
copper(II) sulfate pentahydrate = CuSO
∙5H
O
4
2
11
Undeca
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2