Questions On Ph Curves
ADVERTISEMENT
C h e m g u i d e – q u e s t i o n s
pH CURVES
1. a) What do you understand by the equivalence point of the reaction between sodium hydroxide
solution and dilute hydrochloric acid?
b) Draw the pH curve you would expect to get if you added 1 mol dm
-3
hydrochloric acid to 25 cm
3
of 1 mol dm
-3
sodium hydroxide solution. Mark the position of the equivalence point on the curve.
c) Now draw the pH curve you would expect if you added 1 mol dm
-3
sodium hydroxide solution to
25 cm
3
of 1 mol dm
-3
hydrochloric acid. Mark the position of the equivalence point on the curve.
2. Repeat this for the following combinations of acid and alkali, in each case marking the position of
the equivalence point.
a) Adding 1 mol dm
-3
hydrochloric acid to 25 cm
3
of 1 mol dm
-3
ammonia solution.
b) Adding 1 mol dm
-3
ammonia solution to 25 cm
3
of 1 mol dm
-3
hydrochloric acid.
c) Adding 1 mol dm
-3
ethanoic acid to 25 cm
3
of 1 mol dm
-3
sodium hydroxide solution.
d) Adding 1 mol dm
-3
sodium hydroxide solution to 25 cm
3
of 1 mol dm
-3
ethanoic acid solution.
e) Adding 1 mol dm
-3
ethanoic acid to 25 cm
3
of 1 mol dm
-3
ammonia solution.
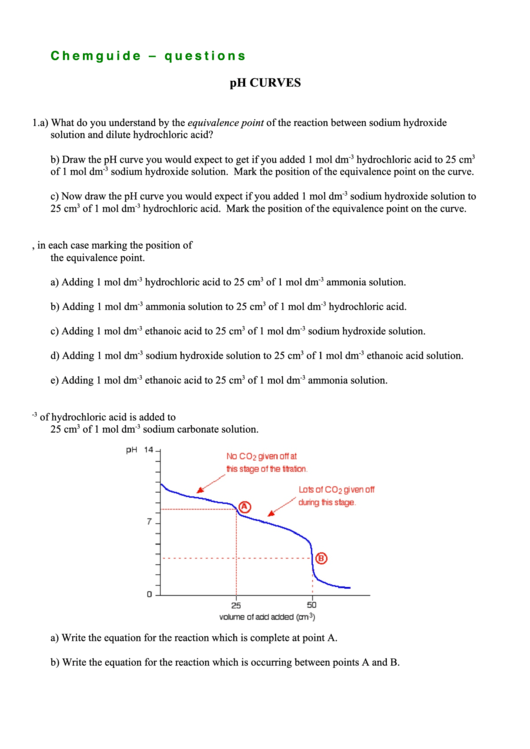
3. The following curve shows the changes in pH when 1 mol dm
-3
of hydrochloric acid is added to
25 cm
3
of 1 mol dm
-3
sodium carbonate solution.
a) Write the equation for the reaction which is complete at point A.
b) Write the equation for the reaction which is occurring between points A and B.
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2








