Chemistry Cheat Sheet
ADVERTISEMENT
7.2
How are Lewis symbols written?
The symbol of an element surrounded by dots, where the number of dots is
equal to the number of valence electrons. Dots are spread out on four sides of
the symbol.
Why do Be, B, and C have 2, 3, and 4 unpaired
Because, when bonding, they act as if they have 2, 3 and 4 unpaired electrons.
electron's according to their Lewis structure, but not
This is also true of other elements in the same groups as Be, B, and C.
according to their electron configurations?
What is a quick way to check the number of valence
Look at the group number of the element. For example, O is in group VI (roman
electrons of a representative element?
numeral 6) and has 6 valence electrons.
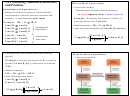
How can the reaction of Na with Cl be represented
+
–
[
]
[
]
Cl
N
a
Cl
Na
+
with Lewis symbols?
How are molecular substances different from ionic
They do not benefit from a lattice energy. Instead, they form because of the
ones?
lowering of energy that comes from sharing electrons.
Between atoms in a molecule, which forces result in
There is an attraction between the nucleus of one atom and the electrons of
attraction and which result in repulsion?
another. There is a repulsion between nuclei of opposite atoms and between
electrons of opposite atoms.
At what distance do atoms remain separated?
At the distance were forces of attraction are equal to forces of repulsion. In
other words, where the energy is lowest.
> Explain why this the lowest energy state?
Because energy would be required to push the atoms closer together. Likewise,
energy would be required to pull them apart.
What is the sharing of electrons called?
An "electron pair bond", or simply a "bond".
How is a bond represented?
With a pair of dots between elements, or with a dash (representing the two
bonding electrons).
7.4-7.7
State the octet rule.
When atoms (other than hydrogen) react, they tend to achieve an outer shell
having eight electrons.
How are double and triple bonds represented?
With 2 or 3 pairs of electrons (or dashes) respectively.
Give examples of molecules that have less than, and
BeCl
and BCl
have less than an octet, PCl
and SF
have more than an octet.
2
3
5
6
more than an octet (2 examples each).
Give the rules for writing Lewis structures.
1) Decide which atoms are bonded
2) add together the number of valence electrons from all atoms
3) subtract two electrons for each bond
4) complete the octet of peripheral atoms (remember, hydrogen has 2 electrons
instead of an octet)
5) place remaining electrons in pairs around the central atom
6) ensure that the central atom has at least an octet, unless it is Be (4 e- instead
of 8) or B (6 e-); form double or triple bonds if necessary.
What is meant by resonance structures? How are
When a molecule cannot be represented adequately by a single Lewis structure.
these drawn?
All contributing structures are drawn, separated by a double headed arrow(s).
Alternatively the resonance bond is represented by a dashed line.
What is commonly misunderstood about resonance
The molecule does not flip-flop between structures. Instead it takes a form that
structures?
is intermediate between the resonance structures.
When are resonance structures drawn?
When a double bond must be formed to give an octet to the central atom, and
there are equivalent choices for the location of the double bond.
7.9
What theory predicts the shape of molecules?
VSEPR (Valence Shell Electron Pair Repulsion) theory
> what is the idea behind this theory?
Bonds contain electrons. These areas of high negative charge will be oriented
such that the distance between them is maximized.
Give the names for the shapes formed when the
2 - Linear (no faces), 3 - Planar triangular (1 face), 4 - Tetrahedral (4), 5 -
number of electron pairs (i.e. bonds) is 2, 3, 4, 5, and
Trigonal bipyramidal (6), 6 - Octahedral (8).
6. Indicate the number of faces/sides of the resulting
structure.
O
What is the bond angle of a tetrahedral molecule?
109.5
O
How can the bond angles for other shapes be
By simple geometry: a circle is 360
, thus a planar triangular molecule (three
O
determined?
bonds) will have bond angles of 120
(360/3 = 120)
What is another name for a pair of electrons that are
Lone pairs.
not involved in bonding?
Give the names for the shapes formed when lone
Tetrahedron (4 bonds, 0 lone pairs) goes to trigonal pyramidal (3,1), then to
pairs are present. (see fig. 7.7 - 7.9 )
nonlinear/bent (2,2).
Trigonal bipyramidal (5,0) goes to unsymmetrical tetrahedron (4,1), T-shaped
(3,2), and linear (2,3).
Octahedral (6,0) goes to square pyramidal (5,1) and square planar (4,2).
How do double and triple bonds affect the shapes of
A double or triple bond acts the same as a single bond when determining the
molecules?
shape of a molecule. Thus, it is somewhat misleading to identify the shape of
an atom based on the number of bonds.
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1