Enthalpy Of Reaction Worksheet Page 4
ADVERTISEMENT
Enthalpy Of Reaction (∆
H)
rxn
CHEM1101
2009-N-12
November 2009
Marks
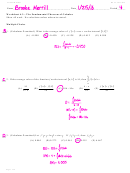
Pentane, CH
CH
CH
CH
CH
, burns completely in oxygen to form CO
(g) and H
O(g). Use the
3
2
2
2
3
2
2
3
atomization enthalpies given below to calculate the enthalpy change for this process.
–1
–1
Δ
H (kJ mol
)
Δ
H (kJ mol
)
atom
atom
pentane
6352
CO
1608
2
O
498
H
O
926
2
2
Answer:
CHEM1101
2007-J-6
June 2007
Marks
The current “petrochemical economy” is based on the combustion of fossil fuels, of which octane
5
is a typical example.
(g) 16CO
2C
H
(l) + 25O
(g) + 18H
O(l)
8
18
2
2
2
Calculate the heat of combustion of octane using the supplied heat of formation data.
–1
–1
–1
Data: C
H
(l): –249.9 kJ mol
; CO
(g): –393.5 kJ mol
; H
O(l): –285.8 kJ mol
8
18
2
2
Answer:
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4