Compounds Handout Key Worksheets With Answers Page 6
ADVERTISEMENT
p. 11
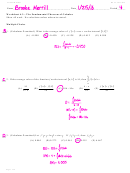
Give the formulas for each of the following molecular compounds:
nitrogen trichloride
dibromine heptaoxide
dinitrogen pentasulfide
NCl
Br
O
N
S
3
2
7
2
5
Name each of the following ions, and determine the formula and name of the
corresponding acid that forms from the ion.
Name of Ion
Formula of Acid
Name of Acid
–
⇒ ⇒ ⇒ ⇒
Cl
=
= _____chloride ion_____
____HCl____ (aq)
___hydrochloric acid____
–2
carbonate ion
H
CO
= carbonic acid
⇒ ⇒ ⇒ ⇒
CO
=
2
3
3
(aq)
–2
sulfite ion
H
SO
= sulfurous acid
⇒ ⇒ ⇒ ⇒
SO
=
2
3
3
(aq)
–3
phosphate ion
H
PO
= phosphoric acid
⇒ ⇒ ⇒ ⇒
PO
=
3
4
4
(aq)
–
nitrate ion
HNO
= nitric acid
⇒ ⇒ ⇒ ⇒
NO
=
3
3
(aq)
CHEMISTRY Naming Compounds Handout Key v 1-3
page 6 of 7
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7