Kinetic Theory Of Matter Worksheet Page 4
ADVERTISEMENT
Use the phase change diagram and your experience with the different states of matter to complete the
table below
Releases
Absorbs
Heat
Heat
Energy
Phase Change
Phase at Start
Phase at End
Energy to
When
i t
Happen
Happens
Ex. freezing
Liquid
Solid
X
1. melting
2. condensation
3. deposition
4. sublimation
5. vaporization
Calculate the following temperatures.
o
6. 10
C = ____________ K
o
7. 0
C = _____________ K
o
8. 0 K = _____________
C
o
9. 100 K = ___________
C
o
10. -40
C = __________ K
11. 285 K = __________ K
Identify each of the following phase changes
12. Ice into a liquid
13. Water vapor into a liquid
14. Solid carbon dioxide into a gas
15. Liquid nitrogen into a gas
16. Iodine gas into a solid
17. Liquid benzene into a solid
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1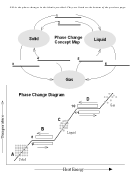 2
2 3
3 4
4