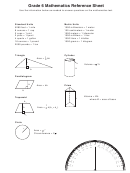
Chemistry Math Reference Sheet Page 3
ADVERTISEMENT
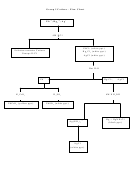
POLAR vs. NONPOLAR MOLECULES
Use the following chart to determine if a molecule is polar or nonpolar.
Note: if the molecule only contains 2 atoms, stop after the first question.
1. Does the molecule have ANY polar bonds?
YES
NO
2. Is the electronegativity of all terminal atoms the
NO
YES
The molecule is NONPOLAR
The molecule is POLAR
3. Is the molecule “symmetrical”?
(Lone pairs on central atom means
molecule is asymmetrical)
NO
The molecule is POLAR
YES
The molecule is
3
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3