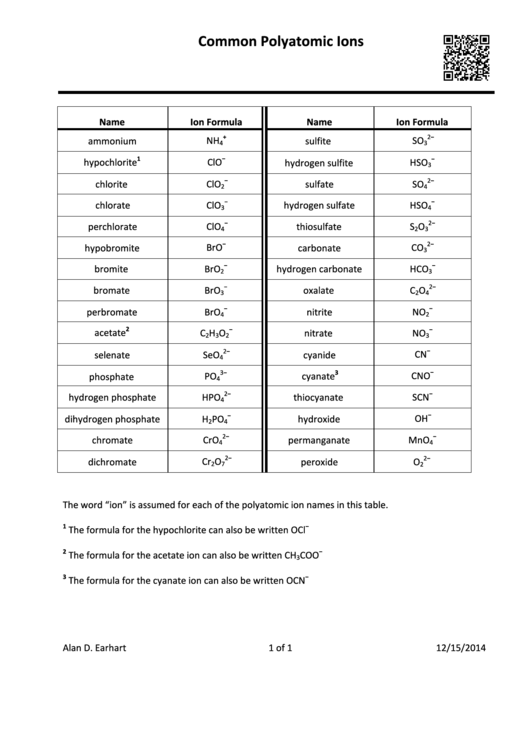
Common Polyatomic Ions Chart
ADVERTISEMENT
Common
P olyatomic
I ons
Name
Ion
F ormula
Name
Ion
F ormula
+
2−
ammonium
NH
sulfite
SO
4
3
−
1
−
hypochlorite
ClO
hydrogen
s ulfite
HSO
3
−
2−
chlorite
ClO
sulfate
SO
2
4
−
−
chlorate
ClO
hydrogen
s ulfate
HSO
3
4
−
2−
perchlorate
ClO
thiosulfate
S
O
4
2
3
−
2−
hypobromite
BrO
carbonate
CO
3
−
−
bromite
BrO
hydrogen
c arbonate
HCO
2
3
−
2−
bromate
BrO
oxalate
C
O
3
2
4
−
−
perbromate
BrO
nitrite
NO
4
2
2
−
−
acetate
C
H
O
nitrate
NO
2
3
2
3
−
2−
CN
selenate
SeO
cyanide
4
−
3−
3
phosphate
PO
cyanate
CNO
4
−
2−
SCN
hydrogen
p hosphate
HPO
thiocyanate
4
−
−
dihydrogen
p hosphate
H
PO
hydroxide
OH
2
4
2−
−
chromate
CrO
permanganate
MnO
4
4
2−
2−
dichromate
Cr
O
peroxide
O
2
7
2
The
w ord
“ ion”
i s
a ssumed
f or
e ach
o f
t he
p olyatomic
i on
n ames
i n
t his
t able.
1
−
T he
f ormula
f or
t he
h ypochlorite
c an
a lso
b e
w ritten
O Cl
2
−
T he
f ormula
f or
t he
a cetate
i on
c an
a lso
b e
w ritten
C H
COO
3
3
−
T he
f ormula
f or
t he
c yanate
i on
c an
a lso
b e
w ritten
O CN
Alan
D .
E arhart
1
o f
1
12/15/2014
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1








