ZIKA TESTING AND NOTIFICATION
2
IgM
RT-PCR
Call to Notify
1-
serology
(serum,
Public Health
Indications for Zika testing
(serum,
urine, or
of these
CSF)
other
special cases
sample
213-240-7941
types)
Pregnancy-associated
Symptomatic pregnant traveler (At least one of the
YES
YES
NO
following: acute onset of fever [measured or reported],
maculopapular rash, arthralgia, conjunctivitis)
3
Asymptomatic pregnant traveler
YES
NO
NO
Pregnant traveler with ultrasound evidence of fetal
YES
YES
YES
rd
microcephaly (occipitofrontal circumference <3
4
percentile for age and gender) and/or calcifications
5
OR fetal loss
7
Infant with microcephaly and/or calcifications and
YES
YES
YES
evidence of maternal Zika virus infection
6
Infant with no apparent defect and evidence of
YES
YES
YES
6
maternal Zika virus infection
Symptomatic pregnant woman without travel history
YES
YES
YES
who had unprotected sex with a symptomatic male
traveler from Zika affected area
In Non–Pregnant Patients
Symptomatic non-pregnant traveler (male or female)
YES
YES
NO
(At least one of the following: acute onset of fever
[measured or reported], maculopapular rash, arthralgia,
conjunctivitis)
Traveler with Guillain-Barré Syndrome diagnosis
YES
NO
YES
For those symptomatic, collect serum for IgM ≥4 days post symptom onset
1
2
If <7 days from onset of symptoms, submit both urine and serum specimens; urine should be collected within 14 days of symptom onset
to improve sensitivity of diagnosis, however urine specimens collected within 30 days will continue to be accepted.
3
Collect sample between 2-12 weeks of return
4
Consider testing amniotic fluid
5
Additional specimens will be requested: e.g. placenta, fetal tissues
6
Positive or inconclusive Zika virus serology
7
Additional specimens will be requested: e.g. cord blood, placenta/umbilical cord tissue, CSF
ZIKA VIRUS TESTING AND REPORT FORM AND INSTRUCTIONS - ZikaInfoTestReq (6/23/16)
Page 5 of 6
CONFIDENTIAL – This material is subject to the Official Information Privilege Act
 1
1 2
2 3
3 4
4 5
5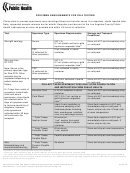 6
6








