Kinetics Worksheet With Answers Page 3
ADVERTISEMENT
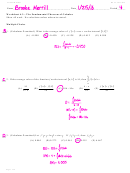
12. Given the reaction:
13. The potential energy diagram below represents the
reaction
S(s) + O
(g) - SO
(g) + energy
2KClO
→ 2KCl + 3O
.
2
2
3
2
Which diagram best represents the potential energy
changes for this reaction?
1)
2)
Which numbered interval on the diagram would change
when a catalyst is added?
1) 1
3) 3
2) 2
4) 4
14. Which information about a chemical reaction is provided
3)
by a potential energy diagram?
1) the oxidation states of the reactants and products
2) the average kinetic energy of the reactants and
products
3) the change in solubility of the reacting substances
4) the energy released or absorbed during the reaction
15. In the potential energy diagram below, which letter
represents the potential energy of the activated complex?
4)
1) A
3) C
2) B
4) D
16. In a potential energy diagram, the difference between the
potential energy of the products and the potential energy
of the reaction is equal to the
1) heat of reaction
2) entropy of the reaction
3) activation energy of the foward reaction
4) activation energy of the reverse reaction
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9 10
10