Ammonia Acid Worksheet With Answers Page 44
ADVERTISEMENT
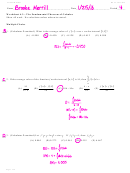
(b)
(i)
reversible reaction (or explanation)
1
(ii)
add water
do not accept cooling or reverse the reaction
1
[3]
M17.
(a)
the bag gets cold because heat energy is taken in from the surroundings
1
(b)
endothermic
1
(c)
any two from:
•
mix / spread (the ammonium nitrate and water)
•
dissolve faster(*)
•
get cold faster or so the whole bag gets cold(*)
(*)allow increase rate or quicker reaction
•
particles collide more or more collisions
2
[4]
M18.
(a)
gives out (heat)
1
(b)
D
1
(c)
L
1
(d)
magnesium chloride
1
[4]
M19.
(a)
(i)
any one from:
•
they are positive / cations
+
•
they are H
•
opposite charges attract
ignore atom
1
Page 44 of 49
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20 21
21 22
22 23
23 24
24 25
25 26
26 27
27 28
28 29
29 30
30 31
31 32
32 33
33 34
34 35
35 36
36 37
37 38
38 39
39 40
40 41
41 42
42 43
43 44
44 45
45 46
46 47
47 48
48 49
49