Chemistry Worksheets
ADVERTISEMENT
Moles 2 – Reacting mass answers
Moles 2 – Reacting mass answers
1)
What mass of hydrogen is produced when 192 g of
1)
What mass of hydrogen is produced when 192 g of
magnesium is reacted with hydrochloric acid?
(3)
magnesium is reacted with hydrochloric acid?
(3)
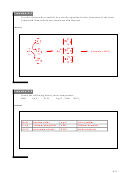
2HCl + Mg → MgCl
2HCl + Mg → MgCl
A)
+ H
A)
+ H
2
2
2
2
Ratio
1
1
Ratio
1
1
A
24
2
A
24
2
R
R
Moles = Mass/A
192/24 = 8 moles
8 moles (As 1:1 ratio)
Moles = Mass/A
192/24 = 8 moles
8 moles (As 1:1 ratio)
R
R
Mass of H
= moles x M
= 8 x 2 = 16g
Mass of H
= moles x M
= 8 x 2 = 16g
2
R
2
R
4)
What mass of iron is formed from 1 kg of iron oxide?
4)
What mass of iron is formed from 1 kg of iron oxide?
+ 2 Al → 2 Fe + Al
+ 2 Al → 2 Fe + Al
A)
Fe
O
O
(3)
A)
Fe
O
O
(3)
2
3
2
3
2
3
2
3
Ratio
1
2
Ratio
1
2
M
160
56
M
160
56
R
R
Moles = Mass/M
Moles = Mass/M
R
R
= 1000g/160 = 6.25moles Fe = 12.5 moles (as 1:2 ratio)
= 1000g/160 = 6.25moles Fe = 12.5 moles (as 1:2 ratio)
Mass of Fe = moles x A
= 12.5 x 56 = 700g (0.7kg)
Mass of Fe = moles x A
= 12.5 x 56 = 700g (0.7kg)
R
R
6)
What mass of aluminium oxide produced when 135 g of
6)
What mass of aluminium oxide produced when 135 g of
aluminium is burned in oxygen?
(3)
aluminium is burned in oxygen?
(3)
→ 2Al
→ 2Al
A)
4Al + 3O
O
A)
4Al + 3O
O
2
2
3
2
2
3
Ratio
4
2
Ratio
4
2
M
27
102
M
27
102
R
R
Moles = Mass/M
Moles = Mass/M
R
R
= 135g/27 = 5
Therefore = 2.5 (as 4:2 ratio)
= 135g/27 = 5
Therefore = 2.5 (as 4:2 ratio)
Mass of Fe = moles x A
= 2.5 x 102 = 255g
Mass of Fe = moles x A
= 2.5 x 102 = 255g
R
R
8)
What mass of hydrogen is needed to react with 32 g of
8)
What mass of hydrogen is needed to react with 32 g of
→ Cu + H
→ Cu + H
copper oxide? CuO + H
O
(3)
copper oxide? CuO + H
O
(3)
2
2
2
2
Ratio
1
1
Ratio
1
1
M
80
2
M
80
2
R
R
Moles = 32/80 =
0.4
0.4 (as 1:1)
Moles = 32/80 =
0.4
0.4 (as 1:1)
Mass of H
needed = moles x A
= 0.4 x 2 = 0.8g (not a lot!)
Mass of H
needed = moles x A
= 0.4 x 2 = 0.8g (not a lot!)
2
R
2
R
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1