Chemistry I Atomic Bonding Practice Quiz With Answer Key - Mr. Scott Page 11
ADVERTISEMENT
56)
Multiple covalent bonds may occur in atoms that contain carbon, nitrogen, or
a) chlorine.
b) hydrogen.
c) oxygen.
d) helium.
57)
The substance whose Lewis structure shows three covalent bonds is
a) H
O.
b) CH
Cl
.
2
2
2
c) NH
.
d) CCl
.
3
4
58)
How many double bonds are in the Lewis structure for hydrogen fluoride, HF?
a) none
b) one
d) three
c) two
59)
How many extra electrons are in the Lewis structure of the phosphate ion, PO
3–
?
4
a) 0
b) 2
c) 3
d) 4
60)
How many electrons must be shown in the Lewis structure of the hydroxide ion, OH
–
?
a) 1
b) 8
c) 9
d) 10
61)
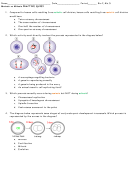
What is the Lewis structure for hydrogen chloride, HCl?
a) A
b) B
c) C
d) D
- 11 -
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20 21
21 22
22 23
23 24
24 25
25 26
26 27
27 28
28 29
29 30
30 31
31 32
32 33
33 34
34