Chemistry I Atomic Bonding Practice Quiz With Answer Key - Mr. Scott Page 23
ADVERTISEMENT
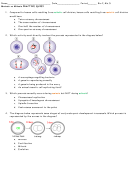
124)
The hybridized orbitals responsible for the shape of the CH
molecule are
4
a) 1s
1
1p
3
.
b) sp
2
.
c) 2s
2
2p
2
.
d) sp
3
.
125)
The mixing of two or more atomic orbitals of similar energies on the same atom to
produce new orbitals of equal energies is called
b) malleability.
a) VSEPR theory.
c) hybridization.
d) dipole-dipole interaction.
126)
Orbitals of equal energy produced by the combination of two or more orbitals in the same
atom are called
b) hybrid orbitals.
a) q-orbitals.
d) n-orbitals.
c) Lewis orbitals.
127)
Which hybrid orbitals help explain how methane bonds?
a) sp
3
orbitals
b) sp orbitals
c) pd
3
orbitals
d) df
3
orbitals
128)
Hybridization helps explain molecular bonding when the valence electrons on the
uncombined atoms
a) total more than 8.
b) are in orbitals with different
shapes.
c) total less than 3.
d) are not available for
bonding.
- 23 -
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20 21
21 22
22 23
23 24
24 25
25 26
26 27
27 28
28 29
29 30
30 31
31 32
32 33
33 34
34