Atoms, Molecules, And Ions Worksheet - Chapter 2 Page 8
ADVERTISEMENT
Isotopes:
• Atoms of the same element with different masses.
• Isotopes have different numbers of neutrons, thus
different mass numbers.
11
12
13
14
C
C
C
C
6
6
6
6
Atomic Mass
Atomic and molecular masses
(actually the mass to charge
ratio) can be measured with
great accuracy with a mass
spectrometer.
8
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20 21
21 22
22 23
23 24
24 25
25 26
26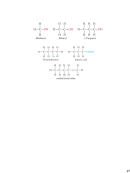 27
27








