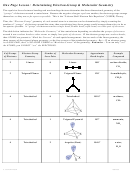
Chem 161 Chapter 9 Molecular Geometry Worksheet - Shape Determines Function Page 15
ADVERTISEMENT
e. CCl
F
Lewis formula
sketch of 3D shape with dipoles
2
2
Molecular geometry: ______________________
CCl
F
is __________.
2
2
Bond angles: ___________________________
(Circle one) polar
nonpolar
f. SOCl
Lewis formula
sketch of 3D shape with dipoles
2
Molecular geometry: ______________________
COCl
is __________.
2
Bond angles: ___________________________
(Circle one) polar
nonpolar
Ex. 3: Rank the following molecules in terms of increasing dipole moment:
CH
Cl
, CCl
F
, CH
F
,and CF
.
2
2
2
2
2
2
4
_____________ < _____________ < _____________ < _____________
smallest dipole moment
largest dipole moment
Ex. 4: Rank the following molecules in terms of increasing dipole moment:
COCl
, SO
, SOCl
, NF
,and NCl
.
2
3
2
3
3
_____________ < _____________ < _____________ < _____________ < _____________
smallest dipole moment
largest dipole moment
CHEM
1 61:
C hapter
9
page
1 5
o f
4 0
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9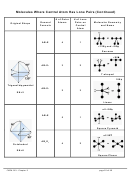 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20 21
21 22
22 23
23 24
24 25
25 26
26 27
27 28
28 29
29 30
30 31
31 32
32 33
33 34
34 35
35 36
36 37
37 38
38 39
39 40
40