Chem 161 Chapter 9 Molecular Geometry Worksheet - Shape Determines Function Page 22
ADVERTISEMENT
–
3
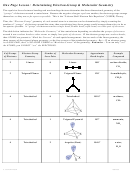
To maximize the distance between e
’s in C, the four sp
orbitals in the C atom are oriented to be 109.5˚ away from one
another in three dimensions
tetrahedral shape (with 109.5˚ bond angles)
→
3
corresponding to four sp
orbitals oriented towards
the four corners of a 4-sided pyramid
3
The four hybrid sp
atomic orbitals for C are shown in blue below,
along with the four 1s orbitals for the four H atoms to form the
CH
molecule.
4
The C-H bonds are made from the electrons shared in the darker overlapping regions.
Case #4: PF
:
5
1. Draw the Lewis formula for the PF
molecule below:
5
Lewis formula
Sketch of molecule
2. PF
’s shape is ____________________________ and its bond angles are ____________.
5
Sketch the molecule above.
3. Electron configuration for P (using core notation): ___________________
CHEM
1 61:
C hapter
9
page
2 2
o f
4 0
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20 21
21 22
22 23
23 24
24 25
25 26
26 27
27 28
28 29
29 30
30 31
31 32
32 33
33 34
34 35
35 36
36 37
37 38
38 39
39 40
40