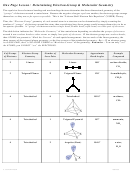
Chem 161 Chapter 9 Molecular Geometry Worksheet - Shape Determines Function Page 40
ADVERTISEMENT
2
2a. Draw the resonance structures for carbonate ion, CO
.
3
−
resonance structure
Sketch of molecule
2
b. CO
’s shape is _____________________________ and its bond angles are __________.
3
−
c. Sketch the molecule above.
2
d. Sketch the 3D arrangement of atoms in the carbonate ion, CO
using ball-and-stick
3
−
representations, then draw the following:
i. The unhybridized p orbitals for the C atom and the three outer O atoms and the resulting
MOs holding the delocalized electrons—i.e., electrons shared by all four atoms
ii. The σ and π bonds in the polyatomic ion
CHEM
1 61:
C hapter
9
page
4 0
o f
4 0
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9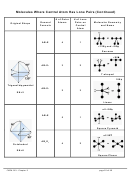 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20 21
21 22
22 23
23 24
24 25
25 26
26 27
27 28
28 29
29 30
30 31
31 32
32 33
33 34
34 35
35 36
36 37
37 38
38 39
39 40
40