Chem 161 Chapter 9 Molecular Geometry Worksheet - Shape Determines Function Page 9
ADVERTISEMENT
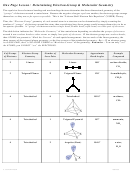
From Octahedral
AB
E: square pyramidal (central atom + 5 B atoms make 4-faced pyramid)
5
– start with AB
(octahedral) and replace one B atom with one lone pair
6
– since all six outer positions are identical, doesn't matter which outer atom you take
→ square pyramidal shape
– bond angles are now less than 90°
AB
E
: square planar (central atom + 4 B atoms form square all in 1 plane)
4
2
– start with AB
(octahedral) and replace 2 B atoms with 2 lone pairs
6
– doesn't matter which B atom taken first, second B atom taken 180° away from
it to maximize space between lone pairs
→ square planar shape
– bond angles are now exactly 90° since lone pairs balance each other
CHEM
1 61:
C hapter
9
page
9
o f
4 0
ADVERTISEMENT
0 votes
Related Articles
Related forms
Related Categories
Parent category: Education
 1
1 2
2 3
3 4
4 5
5 6
6 7
7 8
8 9
9 10
10 11
11 12
12 13
13 14
14 15
15 16
16 17
17 18
18 19
19 20
20 21
21 22
22 23
23 24
24 25
25 26
26 27
27 28
28 29
29 30
30 31
31 32
32 33
33 34
34 35
35 36
36 37
37 38
38 39
39 40
40